Blood pressure devices are used in clinics, hospitals, and sometimes at home to measure how hard your blood pushes against your artery walls. Many people want to know if their device is accurate and safe. One term you might see is “FDA-cleared.”
This guide explains what FDA-cleared blood pressure devices are, what medical-grade monitoring means, and how doctors use these tools in real medical care.
This article is for education only and does not recommend any specific product or treatment.
What Does “FDA-Cleared” Mean for Blood Pressure Devices?
In the United States, medical devices are regulated by the U.S. Food and Drug Administration (FDA). The FDA checks that devices meet certain safety and performance standards before they are sold.
Most blood pressure monitors are reviewed through a process called 510(k) clearance. In this process, a company must show that its device is similar to another device already legally on the market.
This means the device is considered “substantially equivalent” to an existing product in how it works and what it is used for.
However, FDA clearance does not mean:
- The FDA tested the device on patients for clinical outcomes
- The device guarantees health improvements
- The device replaces a doctor’s judgment
Instead, FDA clearance mainly confirms that the device meets regulatory safety and performance standards.
In simple terms:
FDA clearance shows the device passed regulatory review, but it does not prove medical benefit for every patient.
What Is a Medical-Grade Blood Pressure Device?
A medical-grade blood pressure device is built for use in clinical or clinician-guided settings. These devices usually go through special testing to confirm they meet strict accuracy standards.
Medical-grade devices are often:
- Tested using formal clinical validation protocols
- Designed to give reliable readings
- Used in situations where readings may help guide medical decisions
Doctors rely on these measurements when they evaluate symptoms, review patient health history, or adjust treatments.
Not every blood pressure monitor sold for home or wellness use meets medical-grade standards.
The term medical-grade usually refers to:
- Validation testing
- Accuracy requirements
- Suitability for clinical use
It does not simply mean better marketing or branding.
Medical-Grade vs Consumer Devices
Blood pressure monitors are sold for different purposes. Some are made for clinical care, while others are meant for personal wellness tracking.
Here is a simple comparison.
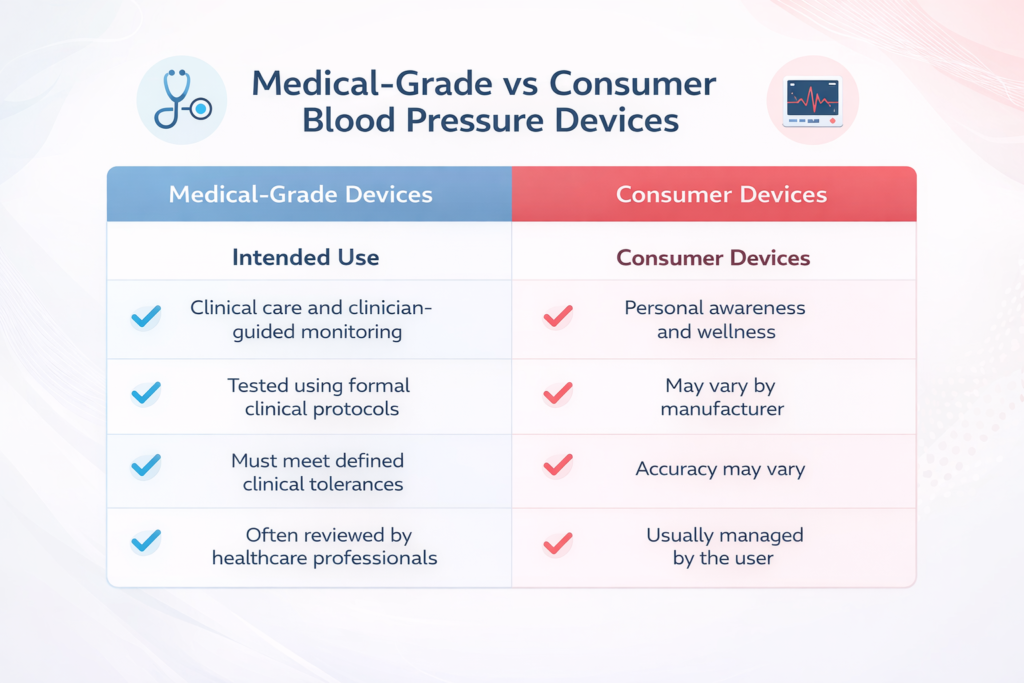
Consumer devices can still help people stay aware of their blood pressure, but they may not always be appropriate for medical decision-making.
How Accurate Are Home Blood Pressure Devices?
Home blood pressure monitors can be helpful, but accuracy depends on several factors.
Even FDA-cleared devices can give incorrect readings if they are used the wrong way.
Common factors affecting accuracy include:
- Using the wrong cuff size
- Holding the arm in the wrong position
- Moving or talking during measurement
- Poor device validation
- Older devices that have worn out over time
Doctors often review home readings along with other medical information, such as symptoms and medical history.
Because of this, a single reading is rarely used by itself to make a major medical decision.
Why Some Blood Pressure Readings Are Incorrect
Blood pressure readings can sometimes be wrong due to technique or environment.
Common causes of inaccurate readings include:
- Incorrect cuff placement
- Sitting in the wrong position
- Talking or moving during the test
- Stress or anxiety
- Using an unvalidated device
Even small mistakes can change the numbers. That is why doctors often recommend taking multiple readings over time.
Learning proper measurement technique can make a big difference in getting reliable blood pressure data.
How FDA-Cleared Blood Pressure Devices Are Used in Clinical Care
Doctors use FDA-cleared blood pressure monitors in several ways.
These include:
- In-office measurements during medical visits
- Remote monitoring supervised by clinicians
- Long-term tracking of blood pressure trends
Healthcare professionals review the data carefully. They look at readings along with:
- Patient symptoms
- Medical history
- Current medications
- Other test results
Blood pressure devices can support medical care, but they do not replace professional medical judgment.
When Blood Pressure Accuracy Matters Most
Accurate readings are especially important in certain medical situations.
For example, doctors may rely on blood pressure measurements when they:
- Adjust medications
- Monitor chronic conditions
- Evaluate treatment response
- Check for possible health risks
If readings are inaccurate, it may lead to unnecessary treatment changes or missed health concerns.
That is why clinicians often emphasize validated devices and proper measurement technique.
Important Notes About This Information
This article is provided for educational purposes only.
It does not:
- Recommend specific blood pressure devices
- Compare brands or manufacturers
- Guarantee health outcomes
- Replace medical advice from a healthcare professional
- Determine eligibility for medical care
Always consult a qualified healthcare provider if you have questions about blood pressure monitoring or medical treatment.




